Paul Wendelboe
Research Assistant & Programmer
Roseville, California, USA
My History & Profile
I'm a current research assistant with the Tantillo group. I have been working on theoretical organic chemistry for the last 4 years in the group ever since I was an undergraduate at UC Davis. I graduated in 2016 with a B.Sc. in pharmaceutical chemistry. During this time I have been working with both chemistry and programming related fields. I have worked on several papers, one of which is still in the process of being completed.
Since I have graduated I have two published papers and I have been learning more about both the chemistry that makes our calculations work as well as the computing processes needed to carry out our calculations. We utilize HPC methods in order to calculate the geometry and energies of molecules of interest. Initially I worked with Prof. T. Andrew Mitchell from Illinois State University. We investigated the mechanism of formation of a five-member ring proceeding through an oxidopyrylium [5+2] intermediate. The resulting calculations helped to shed light on the reaction mechanism as well as show the probable rate determining step thru calculation and matching of the kinetic isotope effect studies originally done by Prof. Mitchell.
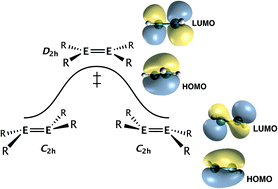
Currently I have been working with Prof. Phil Power in order to investigate the second order and pseudo second order Jahn-Teller effects in various analogues of Ammonia, Ethylene, and Acetylene. We have one published tutorial on this in organometallics, and we except further papers in this area. The chemistry at work in this is rather interesting as it shows the effect that symmetry can have on the structure of heavier analogues of Ethylene and Acetylene. Rather than being linear or planar as the carbon versions are, we see a bending in the heavier analogues and the effect is nearly entirely attributable to symmetry effects on the molecule. By breaking symmetry you can lower the overall energy of the orbitals and thus have a lower energy structure.
During all this time I have also been working on improving my programming skills. During the summers of 2011 to 2013 I worked with the company Productive Solutions on RFID based bus passes. I used the language C# in order to create and modify the RFID cards that are used by several major transportation companies around the United States. I was able to initialize a card from scratch according to the protocol that was established by the company and was able to provide technical support and my own expertise when it was needed in order to meet deadlines and ship code and projects on time. The RFID cards used were produced by MiFare, however I'm unable to comment on the exact types at this time. This environment showed me the importance of delivering code on time and correct the first time, as once it is in the production environment there would be minimal ability for us to change it. As such I delivered consistent code according to a pre-defined specification as well as ever evolving business requirements.
Due to the start of my undergraduate research in 2014 I was unable to continue to work with Productive Solutions, however I have been improving my code skills with small projects as well as improving my scripting skills in order to analyze the data that is generated in the course of our calculations. This has lead to me understanding scripting in both languages such as Python as well as using the command line tools that BASH provides. This has enabled me to speed up processing data and in general while the calculations can take days to complete, the analysis of it can take minutes. I have also been learning more about low level programming and have even implemented a SHA256 hashing algorithm that can, at the extreme, calculate up to 8 SHA256 hashes at once using the AVX SIMD instruction set. This project will be posted soon, as I would like to clean it up a little before releasing it to the world. As a side note however because it was merely a learning project for me I wouldn't use it in production code as a hashing algorithm should ideally be time independent and I'm unsure if mine is.
Skillset
Development
- Python
- C#
- C/C++
- X86 assembly
- X86_64 assembly
- X86 SIMD
- Go
- Some AWS architecture
- Some Java
Software
- MS Office
- Gaussian
- Gaussview
- Visual Studio Code
- Visual Studio 2017
- ChemBioDraw 16
Past Education
Granite Bay High School graduate of 2011.
UC Davis graduate of 2016.
Research Experience
Research Assistant for Dean Tantillo in the Tantillo Group 2014-Present
Research Assistant for Karl Anker Jørgensen at Aarhus Univeristy for the Summer of 2015
Work Experience
Freelance Developer 2011-2013
Publications
Oxidopyrylium-Alkene [5 + 2] Cycloaddition Conjugate Addition Cascade (C3) Sequences: Scope, Limitation, and Computational Investigations • Oxidopyrylium-alkene [5 + 2] cycloaddition conjugate addition cascade (C3) sequences are described. Intramolecular cycloadditions involving terminal alkenes, enals, and enones were investigated. Substrates with tethers of varying lengths delivered five- and six-membered carbocycles and heterocycles thus demonstrating the scope and limitation of the cycloaddition–conjugate addition cascade. Several experiments and theoretical calculations provide evidence for the proposed mechanistic pathway. •Published on July 12, 2018 in The Journal of Organic Chemistry 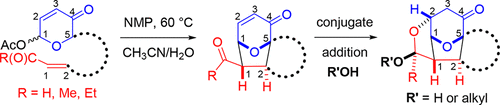
Second-Order Jahn–Teller (SOJT) Structural Distortions in Multiply Bonded Higher Main Group Compounds • The geometrical deviations from planar to folded or from linear to bent structures observed in multiply bonded heavy main group element analogues of ethylene and acetylene are treated from a second-order Jahn–Teller (SOJT) perspective. The structural distortions can be regarded as a consequence of a mixing of the filled highest occupied molecular orbital (HOMO) and an unoccupied orbital (LUMO+1). This mixing stabilizes the HOMO and destabilizes the unoccupied LUMO+1 orbital and produces nonbonding electron density, which causes the geometrical distortion through interelectronic repulsion. This occurs more readily in the heavier species because of their generally weaker bonds, which permit a closer approach and hence greater interaction of the molecular levels. Predicting which geometrical distortion enables the HOMO/LUMO+1 mixing requires knowledge of the symmetries of these frontier orbitals and the symmetry of the distortion. For the undistorted structures the product of the symmetries of the interacting orbitals must correspond to the symmetry of the distortion. More formally, the product of the symmetries of the HOMO, of the distortion (which is one of the fundamental vibrational modes of the molecule), and of the LUMO or LUMO+1 must contain the totally symmetric irreducible representation of the molecular point group. This approach is similar to the SOJT treatment of inversion in trigonal-pyramidal main-group species. Published on September 12, 2018 in Organometallics
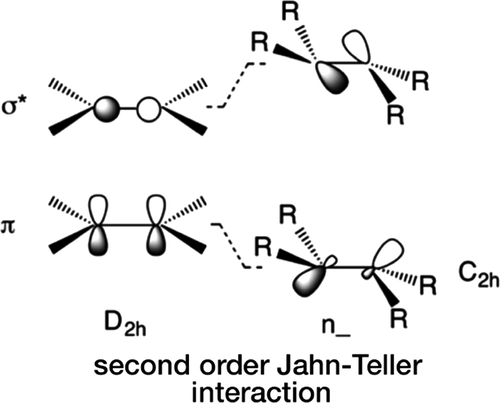
Second order Jahn–Teller interactions at unusually high molecular orbital energy separations • Second order Jahn–Teller (SOJT) effects arise from interactions between filled and empty molecular orbitals of like symmetry. These interactions often lead to structural distortions whose extent is inversely proportional to the energy difference between the interacting orbitals. The main objectives of the work described here are (1) the calculation (using density functional theory methods) of the energies of the valence molecular orbitals in the species EH3 (E = N, P, As or Sb), HEEH (E = C, Si, Ge or Sn), and H2EEH2, (E = C, Si, Ge or Sn) and (2) the correlation of these energies with barriers for planarization or linearization. The calculations suggest an upper limit of about 12 eV energy separation of the interacting levels for SOJT effects to be significant, which is considerably larger than previously thought and implies that SOJT effects may be more common than currently realized. Published on March 20, 2020 in Dalton Transactions